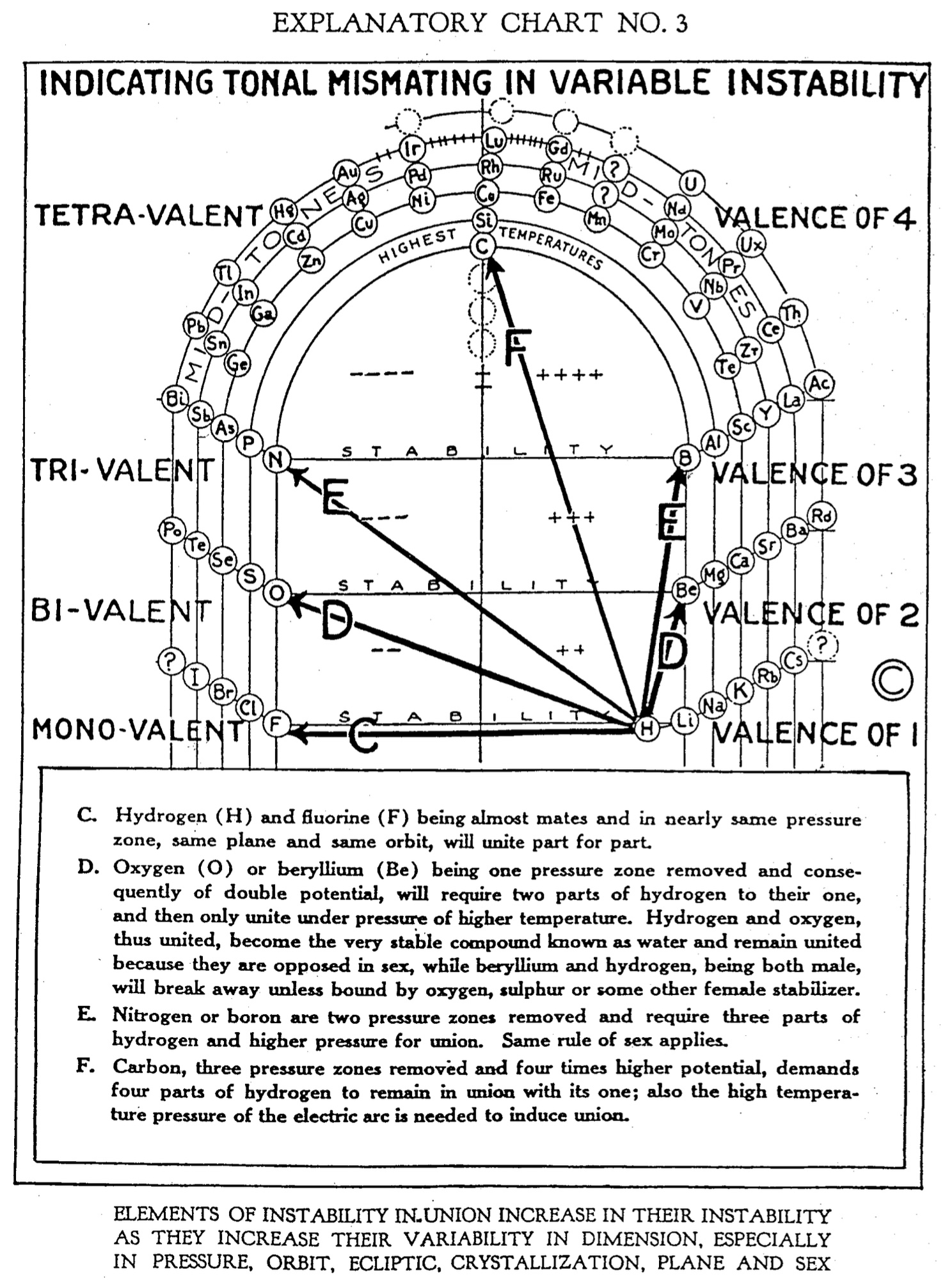
C. Hydrogen (H) and fluorine (F) being almost mates and in nearly same pressure zone, same plane and same orbit, will unite part for part.
D. Oxygen (0) or beryllium (Be) being one pressure zone removed and consequently of double potential, will require two parts of hydrogen to their one, and then only unite under pressure of higher temperature. Hydrogen and oxygen, thus united, become the very stable compound known as water and remain united because they are opposed in sex, while beryllium and hydrogen, being both male, will break away unless bound by oxygen, sulphur or some other female stabilizer.
E. Nitrogen or boron are two pressure zones removed and require three parts of hydrogen and higher pressure for union. Same rule of sex applies.
F. Carbon, three pressure zones removed and four times higher potential, demands four parts of hydrogen to remain in union with its one; also the high temperature pressure of the electric are is needed to induce union.
ELEMENTS OF INSTABILITY IN UNION INCREASE IN THEIR INSTABILITY AS THEY INCREASE THEIR VARIABILITY IN DIMENSION. ESPECIALLY IN PRESSURE, ORBIT, ECLIPTIC, CRYSTALLIZATION, PLANE AND SEX
