As the three planes of gyroscopic rotation compress upon each other they each tend to create a spinning vortex open at each end. Because the centralizing syntropic force was given predominance these vortices will increase in spin velocity the smaller, more compact and more energetic they become. A certain order of Etheric substance is syntropically attractive in that the greater it's velocity the greater is it's mutual attraction upon it's self and others.
"...vortex action (is) induced by differential conflict between the low and high tensions..." Keely
In this wiki we have been saying the above as "vortex motion is caused by the interactions of differential densities..." I think perhaps Keely's definition is better because the term "densities" can be misleading with our readers while "tensions" better explains the multi-dimensional conditions referred to as "...low and high tensions...". Tension refers to the stresses of mutual attraction and dispersion between centers. Differential Densities has been covered in 2.7 - Differential Densities, 16.13 - Differential Densities and Part 12 - Russells Locked Potentials wherein maximum tension is as 4++ level in Russell's Scale of Locked Potentials. Take for instance the air we breathe and have our existence in. This air is a soup of particles being mostly molecules and atoms of oxygen, nitrogen, argon, etc. Likewise it also contains electrons, protons, muons and etherons, among other energetic centralizations (particles). SVP would call this collection as having multi-dimensions - wherein each energy range/band is a different dimension. Each of these entities has a different set of parameters of size, weight, syntropic and entropic balances and energy level. Under a given circumstance each behaves in a certain way specific to its order. Under a given outside influence oxygen particles will behave differently than will electrons or etherons. Therefore introducing a specific influence into a given volume containing a multi-dimensional gas will cause a separation of those dimensions into lock-step actions of the different groups of particles/entities according to their individual parameters. For instance, taking a large volume of still air particles. If a bit of heat is introduced in one part of the volume currents will be set up as heated and partially heated molecules begin to move and displace higher elevated cooler particles. The cooler particles will descend displacing lower warmer particles which then move upwards. The warmer particles push apart into lower tension and density while the cooler ones draw together into higher tension and density. These two streams will tend to curve around each other as they attempt to accomodate each others' different tension (degree of expansion or contraction). Figure 5.0 - Under a common influence different entities move differently according to their Energy State gives an approximation of tension as a function of location within a vortex structure. Figure 5.1 - Swirling Vapor in the Atmosphere shows how masses of cool and warm air "dance" around each other. See Part 12 - Russells Locked Potentials and Addendum 9. As a rule the closer to the electromagnetic center line the higher the tension and density and vice versa, as measured with Russell's Scale of Locked Potentials.
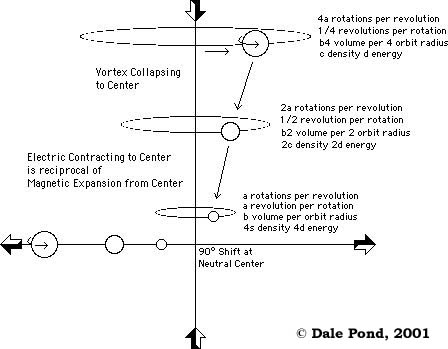
Figure 5.0 - Under a common influence different entities move differently according to their Energy State. (click to enlarge)
In the above image we see how particles rotate in larger orbits thus traveling longer distances than particles closer to the center thus delineating tension, density, etc.. See Part 12 - Russells Locked Potentials for more explanation of Relative and Proportional velocities and energies.
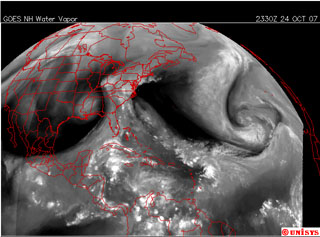
Figure 5.1 - Swirling Vapor in the Atmosphere. (click to enlarge)
In this image we see particles of water vapor acting in concert in huge swirling patterns and flowing currents governed by different tensions.
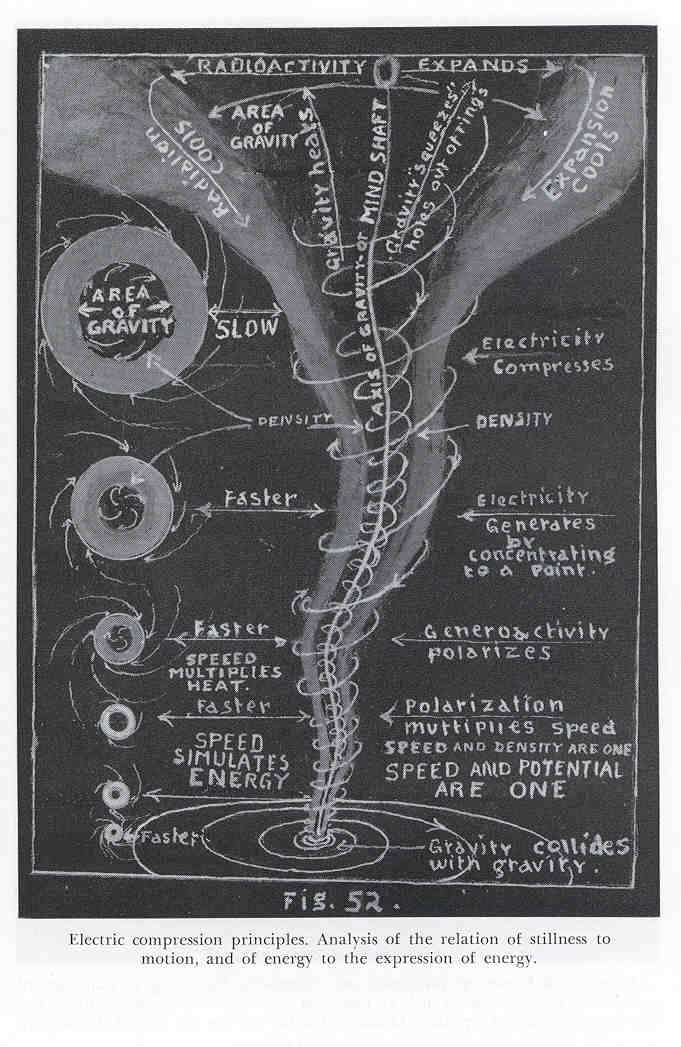
Figure 5.2 - Centers of Swirls are High Tension while Swirl Periphery are Low Tension. (scan courtesy of University of Science & Philosophy) (click to enlarge)
See Also
9.20 - Center and Periphery
Figure 5.2 - Centers of Swirls are High Tension while Swirl Periphery are Low Tension
Figure 9.6 - Center and Periphery
flow
peripheral flow
peripheral mass
periphery
Keely says "All such experiments invariably resulted in vortex motion, whether induced sympathetically or otherwise. All corpuscular action in Nature is vortex motion. The undulatory theory of light is only hypothetical. The conditions of electromagnetic radiation, on the same plane of matter as light, disprove the undulatory theory in many particulars. The vortex action induced by differential conflict between the low and high tensions shows conditions analogous to those in the molecular dissociation of water into hydrogen and oxygen - in other words, vortex action of the highest order but peripheral only. Were it otherwise, the ether could not be held suspended or enclosed in the molecular or atomic envelopes." The Snell Manuscript
See Also
2.7 - Differential Densities 3.6 - Differential Densities Begin to Form 5.1 - Vortex Formation 5.2 - Vortex Motions in Resonators 5.3 - Vortex Motion in Ether and Atoms 5.4 - Vortices are born of Motion and Matter is concentrated Vortices 5.5 - Double Vortices 5.6 - Vortex Forming Spheres 5.9 - Vortices Form Linked Atoms 16.13 - Differential Densities differential densities Figure 13.21 - Differential Pressure Zones in Antagonism cause Rotation On the Partial Differential Equations of Mathematical Physics Vortex
